Achievements, challenges, and future directions
Winter 2025-2026

Download the alternative format
(PDF format, 5.26 MB, 24 pages)
Organization: Office of the Chief Science Advisor
Published: 2026
Table of Contents
- Introduction
- The history of the federal scientific integrity policy in Canada
- Policy implementation – progress and gaps
- Scientific integrity policies in action
- Reflections on 6 years of implementation
- Considerations for strengthening scientific integrity across the federal government
- Looking forward: Strengthening scientific integrity in a changing landscape
- Appendix A: List of departments and agencies that are required to implement a scientific integrity policy
- Appendix B: List of the fifteen scientific integrity policy compliance measures (CMs)
- Appendix C: Summary of initiatives advancing the objectives of the scientific integrity policy, as reported by departments and agencies
Introduction
Governments make policy decisions in response to societal challenges and the needs of the public, and science is an important input into the decision-making process. When informed by robust science, policies are more likely to achieve their objectives. Given its important role, it is essential that science is conducted and communicated in adherence to high standards of scientific integrity – that is, the principles of transparency, openness, rigor, in the absence of real or perceived conflict of interest, impartiality and high ethical standards. When government science is underpinned by scientific integrity, it fosters public trust and reinforces the credibility of both the knowledge it produces and the decisions it informs.
The history of the federal scientific integrity policy in Canada
Development of a model scientific integrity policy
In 2015, the Government of Canada committed to prioritizing science and evidence-based decision-making, stating it is a “government that believes in science – and a government that believes that good scientific knowledge should inform decision-making"Footnote 1. As part of this commitment, and in line with the Minister of Science’s mandate to create a Chief Science Officer “mandated to ensure that government science is fully available to the public, that scientists are able to speak freely about their work, and that scientific analyses are considered when the government makes decisions”Footnote 2, the Government sought advice from the newly appointed Chief Science Advisor on the development of directives and guidelines for the responsible conduct, communication and use of federal science and research.
In July 2018, after a period of extensive consultation and collective efforts, the Model Policy on Scientific Integrity was publishedFootnote 3. Co-developed by the Office of the Chief Science Advisor (OCSA), the Professional Institute of the Public Service of Canada (PIPSC) and the Treasury Board Secretariat (TBS), the policy provides a clear statement to federal researchers and scientists on how they can communicate their work openly, as well as a framework for both employers and employees to ensure the responsible conduct of government science.
Under the Memoranda of Agreements signed on May 15 and June 1, 2017, between TBS and PIPSCFootnote 4, Footnote 5, federal departments and agencies with more than 10 employees within the Research (RE) or Applied Science and Patent Examination (SP) occupational groups were required to adopt or adapt the model policy. The agreements also indicated that the Governance Committee for Implementation of Government-Wide Scientific Integrity (the Governance Committee)Footnote 6, composed of the Secretary of the Treasury Board, the Chief Science Advisor and the President of PIPSC, will meet annually to review progress towards the development and implementation of departmental scientific integrity policies and procedures.
In 2018, TBS issued a notice to heads of human resources identifying 21 departments that employed more than 10 employees in the RE or SP occupational groups, and were therefore required to implement a scientific integrity policy that “apply to all employees who design, conduct, communicate, manage, review or make use of research, science or related activities.”Footnote 7 In addition to these departments, the National Research Council and the Canada Food Inspection Agency were also included. Today, following a number of organizational changes, a total of 25 departments and agencies are required to implement a scientific integrity policy (Appendix A), while a number of other federal organizations are actively exploring the option of voluntarily adopting the policy.
Since 2019, the OCSA has been the primary point of contact for the scientific integrity policy (SIP) and has worked closely with organizations to support and provide advice on policy development and implementation.
Monitoring policy implementation
As per the Model Policy on Scientific Integrity, it is the responsibility of deputy heads to monitor compliance with the requirements of their SIPs, and to report this annually to the Governance CommitteeFootnote 8. To assist deputy heads, the OCSA designed a survey composed of compliance measuresFootnote 9, each of which is associated with a non-discretionary provision of the model policy (Appendix B). The compliance survey has been administered to implicated departments and agencies annually since January 2020 to determine their progress in implementing their SIPs. Results of the surveys are reported on the OCSA website, under the section “The Status of Federal Scientific Integrity Policies”Footnote 10.
Policy implementation – progress and gaps
The 2025 survey was administered at the beginning of the year to the 25 departments and agencies required to have a SIP, all of whom have policies in effect. Together, they cover 2,671 RE employees and 10,821 SP employees, respectively (Figure 1). These numbers represent the minimum population to which departmental SIPs apply. In practice, the policies also extend to employees outside of these occupational groups who are directly or indirectly engaged in research, science or related activities, as outlined in the policy scope.
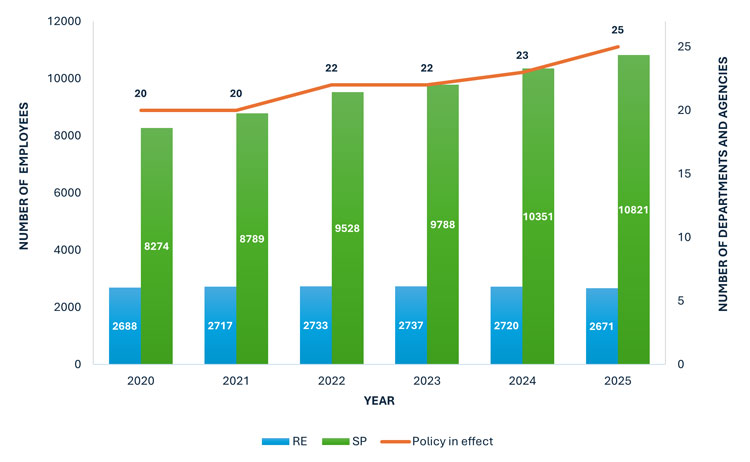
Figure 1: The number of federal departments and agencies that have a SIP in effect, and the total numberFootnote 11 of employees in the research (RE) and applied science and patent examination (SP) occupational groups in each year tracked.
-
Figure 1 - Text description
The combination chart includes a line graph that shows the number of departments and agencies with a scientific integrity policy in effect from 2020 to 2025. This number increased from 20 in 2020 to 25 in 2025. The chart also includes a double bar graph that shows the number of employees in the Research (RE) and Applied Science and Patent Examination (SP) occupational groups within departments and agencies with scientific integrity policies in effect from 2020 to 2025. RE employees increased steadily from 2,688 in 2020 to 2,737 in 2023, and then decreased to 2,671 by 2025. SP employees increased from 8,274 in 2020 to 10,821 in 2025.
Progress in policy implementation among departments and agencies has been good, though variable. To date, 19 of the 25 departments and agencies have reported implementing at least 80% of the compliance measures, with four organizations meeting all requirements applicable to them. Two organizations have made moderate progress, with up to five applicable measures not yet implemented. Four departments and agencies report at least ten applicable compliance measures still outstanding (Figure 2).
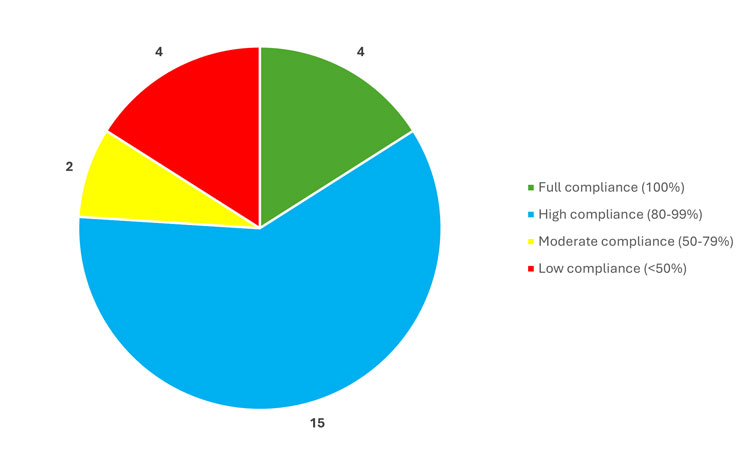
Figure 2: Number of departments and agencies categorized by their level of adherence to applicable compliance measures as of March 31, 2025 – full (100%); high (80-99%); moderate (50-79%); low (less than 50%).
-
Figure 2 - Text description
The pie chart shows the level of compliance with scientific integrity policies among departments and agencies. Four are in full compliance, 15 are in high compliance, two are in moderate compliance, and four are in low compliance.
Progress in policy implementation has also been variable across compliance measures (Figure 3). The measure with the greatest increase in uptake since 2020 is the requirement of notifying and informing contractors or extramural collaborators about the departmental SIP and encouraging them to comply with its provisions (CM7).
Similarly, significant progress has been made in the implementation of measures supporting education, training or professional development in areas such as responsible conduct of research, research ethics and the management, annotation and archiving of research and scientific data (CM13), as well as in the establishment of a mechanism for soliciting advice from researchers and scientists on departmental policies or regulations (CM10).
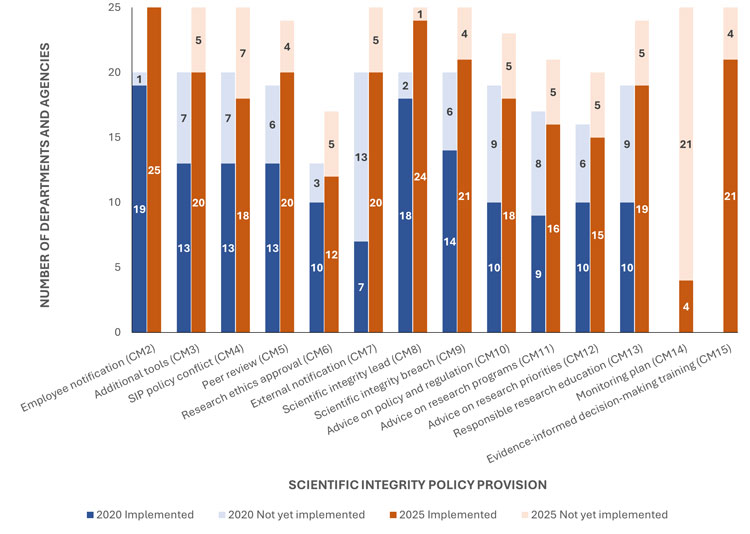
Figure 3: The number of departments or agencies with an approved SIP who have or have not yet implemented the actions or activities corresponding to a particular non-discretionary provision in the model policy as of January 30, 2020 and as of March 31, 2025. For each compliance measure, organizations whose policy does not include the provision in question or that elected not to report on the associated provision have been excluded from the analysisFootnote 12. For a description of the listed compliance measures, see Appendix B.
-
Figure 3 - Text description
The stacked bar graph shows the number of departments and agencies with approved scientific integrity policies that implemented various policy provisions in 2020 and 2025.
In 2025, the provision with the lowest level of implementation is the SIP monitoring plan, with four organizations. The research ethics approval provision is the second lowest, with 10 organizations in 2020 and 12 in 2025. Employee notification is the most widely implemented provision, with 19 organizations in 2020 and 25 in 2025. The appointment of a scientific integrity lead is the second most implemented provision, increasing from 18 organizations in 2020 to 24 in 2025. The largest growth occurred in evidence‑informed decision‑making training, which reached 21 organizations in 2025. No provision showed a decrease in the number of implementing departments and agencies between 2020 and 2025.
In other areas of SIP implementation, departments have been slower to meet the requirements of their policies. For example, for maintaining a record of all instances of conflict or incompatibility between the departmental SIP and other policies, legislation or policy instruments (CM4), and establishing a mechanism to solicit advice from researchers and scientists on departmental research priorities (CM12), only five additional departments became compliant between 2020 and 2025. Another challenging compliance measure is the requirement for departmental research or scientific projects involving humans to be reviewed and approved by a Research Ethics Board (REB) because there is limited REB capacity to support departments without an in-house board; solutions to address this issue are currently being explored.
Finally, the compliance measure with the least progress reported is the development of a performance monitoring plan to assess the effectiveness of departmental SIPs, as outlined in section 7.9 of the model policy. At the time of the 2025 survey, 21 of the 25 departments and agencies had not yet implemented such a plan. Of these 21 organizations, two reported that their performance monitoring plans have been submitted for approval, 12 indicated that their plans were still in development and seven had not yet begun developing a plan (Figure 4). Reasons cited for not developing a plan yet have included limited resources, competing operational demands and priorities, small department size with few employees in the RE and SP categories, internal reorganization and the recent implementation of their SIPs.
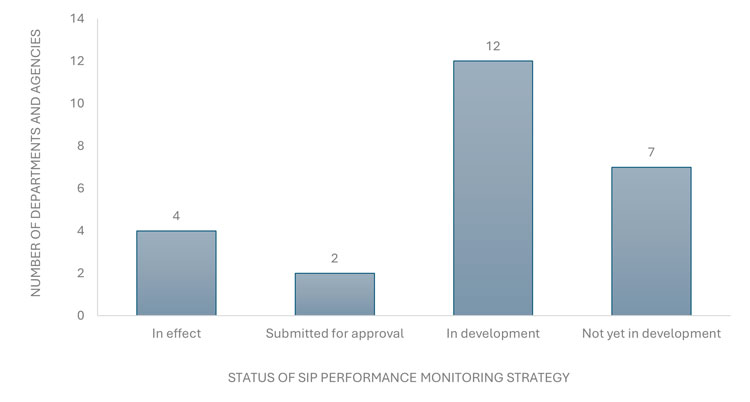
Figure 4: The status of the SIP performance monitoring plan in federal departments and agencies as of March 31, 2025.
-
Figure 4 - Text description
The bar graph shows the status of scientific integrity policy performance monitoring strategies among the 25 departments and agencies with policies in effect. Four departments and agencies reported having an approved strategy in effect, two reported having submitted a strategy for approval, 12 reported developing a strategy, and seven reported having not yet begun developing a strategy.
Scientific integrity policies in action
While departments and agencies have had the flexibility to adapt the model policy to fit their specific organizational contexts, all existing departmental SIPs are very similar, if not identical, to the model policy. Where there has been notable variation is in the implementation approaches taken by different organizations.
One area where differences are observed is in the measures taken to support scientific integrity training and education. Of the 25 organizations surveyed, 18 have made certain trainings related to scientific integrity and ethics mandatory for their employees. These include online, self-paced training on science ethics and integrity, training on the departmental SIP and the Evidence-Informed Decision-Making modulesFootnote 13 co-developed by the OCSA and the Canada School for Public Service (CSPS). Furthermore, some organizations have incorporated scientific integrity into their onboarding workshops and trainings for new employees, linking scientific integrity to departmental values and ethics.
Another notable variation in the implementation of SIPs across departments lies in how scientific integrity leadership positions are structured. While the model policy requires departments and agencies to appoint a Science Integrity Lead (SIL) responsible for addressing allegations of breaches of scientific integrityFootnote 14, this role is filled in a number of different ways across departments. Some departments have designated existing positions – such as their Ombudsperson, Ethics Officer or Integrity Officer – as their SIL, while others have appointed their departmental Chief Scientist or Science Advisor to fill that role. In some cases, senior executives from science branches, including assistant deputy ministers or director generals, have taken on the SIL role.
In addition to managing allegations of policy breach, six departments report that their SIL also plays a broader role in promoting a culture of scientific integrity and ethics in their organization. Beyond the SIL, departments have described a range of approaches and roles that support and promote a culture of scientific integrity, excellence and responsible conduct in research, depending on their structure and mandate. For example, some science-based departments note that the responsibility to foster a culture of scientific integrity is shared across leadership positions such as director generals, directors and managers in science-related portfolios. Others have leveraged their ombuds and integrity offices to promote science excellence and ethics. A few have appointed champions, including at the ADM level, to help reinforce leadership in science.
Departments and agencies are encouraged to share their implementation approaches and best practices at the regular meetings of the Interdepartmental Scientific Integrity Policies Implementation Working Group, of which all 25 organizations, the OCSA, PIPSC and TBS are members. The work and mutual learning undertaken by the working group have helped promote a consistent understanding and interpretation of the policy, enabling diverse implementation strategies that can together strengthen the overall culture of scientific integrity in the government.
Scientific Integrity in Practice: Departmental Spotlights
Across the 25 departments and agencies, initiatives have emerged that reflect and reinforce the core objectives of the Model Policy on Scientific IntegrityFootnote 15. Many of these efforts were developed in response to departmental priorities, unique mandates, and operational contexts. While some initiatives were created directly in response to SIP commitments, others demonstrate how existing or parallel efforts can align with and advance SIP principles and objectives. The following examples illustrate how departments are embedding scientific integrity into internal processes—through learning and training, research management systems, communities of practice, and science governance structures. Together, they highlight the varied and evolving ways in which scientific integrity is being strengthened across the organizations subject to the SIP.
1) Health Canada’s Scientific Integrity Training and Learning Activities
Health Canada has developed learning tools to embed scientific integrity into departmental culture and practice. In 2023, the department launched an in-house Scientific Integrity course that presents key aspects of the Health Canada and Public Health Agency of Canada SIP (HC-PHAC SIP) through engaging scenarios and interactive activities. The course helps apply the policy in day-to-day practice, teaches principles and standards of responsible research conduct, and guides staff on how to report breaches, all with the goal of reinforcing a culture of integrity. This voluntary training is part of the department’s science competency development roadmap, which is promoted through executive performance commitments. Participant feedback has indicated that the course provides a comprehensive overview of the scientific integrity policy, with some recommending that the training be made mandatory. The scenario-based learning components were frequently mentioned as useful by those who completed the training, along with suggestions to further enhance interactivity. Complementing this, Health Canada also developed Strengthening Science-Informed Decision-Making: A How-To Guide, a course for both science and non-science roles that builds skills in evaluating evidence, communicating uncertainty, and engaging in science-policy dialogue. Health Canada leveraged the increased awareness of scientific integrity to strengthen SIP linkages and embed relevant concepts throughout the decision-making training, despite the course not being developed specifically for SIP implementation. Together, these initiatives demonstrate how departmental learning strategies can advance scientific integrity by building capacity across roles and functions.
2) Agriculture and Agri-Food Canada’s Science Management Solution
Agriculture and Agri-Food Canada (AAFC) has used its departmental SIP to guide the evolution of its Science Management Solution (SMS), a centralized system for managing research projects. Beyond planning and tracking, SMS supports governance decisions, project reporting, and documenting research outputs. Although the SMS was not developed specifically in response to the SIP, the policy has significantly influenced how the system functions. SIP principles have shaped improvements in standardized workflows, embedded decision documentation, centralized record-keeping, and streamlined oversight processes. These changes have enhanced transparency, accountability, and collaboration across research teams, while also improving alignment with departmental priorities. By aligning system improvements with SIP principles, AAFC has strengthened its ability to oversee and manage research activities, ensuring they reflect departmental standards for transparency and integrity. Looking ahead, the next generation of SMS will build on this foundation with more intuitive tools and enhanced analytics, further supporting SIP’s objectives.
3) Transport Canada’s Science Community of Practice
Transport Canada’s SIP implementation activities directly informed the development of the department’s Science Community of Practice (CoP), launched in July 2025. Monitoring efforts such as employee surveys and engagement led by the Departmental Science Advisor identified gaps in awareness of SIP principles and opportunities to strengthen science advice and collaboration across the department. These findings helped guide the design of the CoP as a mechanism to address those gaps and promote consistent application of science and technology policies. Since its launch, the CoP has supported improvements in internal processes by increasing SIP awareness, facilitating knowledge exchange through science cafés and lecture series, and enabling resource sharing through an internal collaborative site. It has also promoted continuous learning and strategic alignment across disciplines. The CoP demonstrates how SIP implementation has influenced departmental initiatives that reinforce science advice and contribute to a stronger internal science culture.
4) Environment and Climate Change Canada’s Science Advice Governance Framework and Science and Technology Branch Science Council
Environment and Climate Change Canada (ECCC) has implemented the Science Advice Governance Framework (SAGF) and the Science and Technology Branch (STB) Science Council to strengthen the role and impact of science in departmental decision-making. The SAGF identifies science priorities across program areas, mobilizes efforts to meet operational and regulatory needs, and facilitates expert advice and collaboration to address knowledge gaps. The STB Science Council, launched in 2023, advises senior management on research priorities, supports initiatives such as the ECCC Science Strategy 2024–2029, and promotes scientific dialogue across the Branch. Both instruments are closely aligned with ECCC’s SIP objectives and contribute to a culture of transparency, collaboration, and evidence-informed decision-making. This example illustrates how strong departmental science governance and strategic coordination can actively advance the principles of scientific integrity in practice.
Reflections on 6 years of implementation
Across the 25 departments with a SIP, implementation efforts have led to meaningful progress in putting in place procedures, policies, guidelines and resources to promote the responsible conduct and use of science in government, and to support scientists’ and researchers’ ability to communicate their work to the public. Since the publishing of the model policy in 2018, and the implementation of a SIP in most departments by 2020, departments have taken steps to embed or reinforce scientific integrity in their operations. These developments reflect commitment to the objectives of the policy, nonetheless implementation has also revealed several persistent challenges.
Persistent misunderstanding of the applicability of the policy
The 25 departments and agencies that require a SIP are so required because they have 10 or more employees within the RE or SP groups, per the relevant collective agreements. However, once implemented the policy applies to all “...employees who design, conduct, communicate, manage, review, or make use of [departmental] research, science or related activities”Footnote 16. While all departmental policies include language indicating this broader applicability of the policy, there has been a persistent misunderstanding among some departments as to whether the policy applies only to RE and SP employees or to all employees that conduct or manage research. This has been a recurring issue that both the Governance Committee and the OCSA have repeatedly endeavored to clarify.
Misperceptions around the applicability of the policy may have contributed to various barriers for implementation, including insufficient resourcing. For some departments and agencies that do not count science and research among their core activities, this has at times bred confusion around why they need to have the policy at all, contributing to inertia around implementation.
A related outstanding issue is the reality that there are science, research and related activities, including in the social sciences, being conducted in departments and agencies outside of the 25 captured under the RE and SP collective agreements. Without a coherent, whole-of-government approach to scientific integrity, questions may arise on the quality and reliability of federal research, which in turn carries risks for the government. Furthermore, the protections offered by the policy are not available equally to all federal employees who conduct science and research.
Lack of policy performance monitoring and evaluation
Most of the information on the progress of the SIP derives from the annual compliance survey, the responses for which are supplied by departments and agencies. The survey itself is primarily concerned with whether the required mechanisms or processes have been put in place to facilitate implementation of the policy, rather than the performance of the policy. Each organization is required to develop and implement a policy performance monitoring plan, with appropriate indicators, to determine whether its policy is achieving its stated objectives and track progress over time. As noted earlier, departments and agencies have overall made little progress on performance monitoring, and as a result there is limited knowledge of how effective their SIPs have been.
The lack of performance monitoring also means that a key perspective has been lacking from the implementation process – the employee perspective. Given that a significant portion of the SIP aims to enable or encourage certain activities and behaviours, as well as protect against undue interference, employee feedback (for example, via a survey) is indispensable for evaluating how well the policy is performing. Without this information, it is difficult for organizations to identify where there may be gaps in guidance or resources, where there may be tension between the SIP and other policies, where SIP processes may be ineffective or where course correction may be neededFootnote 17.
To support departments and agencies in fulfilling the requirements for performance monitoring and evaluation, the OCSA developed a model performance monitoring plan, performance indicators and a model employee survey. To date, only one department has reported having administered an employee survey as part of their SIP performance monitoring plan.
Challenges around resources
Many of the policy provisions for which implementation has lagged are those requiring more resources, such as performance monitoring, training and ensuring processes for soliciting researcher or scientist advice. Some provisions, like the requirement for research projects involving humans to be reviewed and approved by an REB, cannot be delivered without the input of appropriate experts. The issue of resources is particularly pertinent for smaller departments and agencies, as well as those who do not house much research or scientific activity, where there may be a lack of capacity or internal expertise.
Considerations for strengthening scientific integrity across the federal government
Scientific integrity policies across all federal organizations
One way to strengthen scientific integrity across the federal government is to make the SIP a mandatory policy for all federal organizations, rather than only the current 25 organizations that are contractually obligated to implement a policy. This could be achieved by, for example, bringing the SIP under a Treasury Board policy instrument, such as the Values and Ethics Code for the Public SectorFootnote 18. A government-wide SIP has a number of benefits. First, it would broaden the applicability of the policy to include all employees involved in science and research activities, irrespective of their classification, ensuring that all federal research activities meet standards of scientific integrity, in line with what is required of academic researchers. Second, given that science is often instrumental in informing policies across all federal departments and agencies, including those that do not conduct in-house science and research activities, a mandatory SIP for all organizations would support advancing a true government-wide culture of scientific integrity. It is noteworthy that, TBS’s 2018 notice to organizations requiring a SIP “encourage[d] all federal departments and agencies to develop and implement Scientific Integrity Policies that apply to ‘all employees who design, conduct, communicate, manage, review or make use of research, science or related activities’”, recognizing the importance of scientific integrity for all of government. Moreover, interest from several federal organizations in adopting a policy voluntarily signals a broader appetite across government for SIPs.
Incorporating scientific integrity into mandatory training for all science-based departments and agencies
Another approach to advancing government-wide scientific integrity is to reinforce training around the policy. Integrating scientific integrity into the onboarding process, as some organizations have already done, for all science-based departments and agencies would amplify awareness and deepen understanding of the policy for all employees who work in organizations with significant science and research activities from the outset. This is the case in post-secondary institutions where researchers are expected to take training modules including in research ethicsFootnote 19. Importantly, the trainings would also draw clear connections between the SIP, departmental codes of conduct and the Values and Ethics Code, clarifying the policy’s relevance and applicability across roles and responsibilities. Furthermore, discussions on how to operationalize the SIP in alignment with the Values and Ethics Code, as well as management of potential intersections between the two, should be part of ongoing training – a need that was identified in the Deputy Minister’s Task Team on Values and Ethics Report to the Clerk of the Privy CouncilFootnote 20.
Bolstering Research Ethics Board capacity
To support cross-government consistency in the ethics review of research involving human participants, improvements to REB capacity within the federal public service should be considered. A centralized or shared services model could be explored to help organizations with limited research activity or resources access qualified experts and receive timely and rigorous review of their research projects as required. A more coordinated approach to REBs could also support the needs of departments and agencies that do not currently have a SIP but do conduct research involving human participants.
SIP performance as part of departmental results
To address the challenges around performance monitoring, the incorporation of SIP performance monitoring into departmental performance measurement and evaluation plans and strategies, as described in Treasury Board’s Policy on ResultsFootnote 21, could be considered. To do so, departments would designate the SIP and its related activities as a program under the Policy on Results’ definition, and apply the same performance measurement and evaluation practices used for other departmental programs. This could also include the development of a set of standardized performance indicators of progress for scientific integrity, based on those developed by the OCSA, to be included in all departmental results indicators. This approach would support more consistent tracking, reporting and continuous improvement of SIP implementationFootnote 22.
Looking forward: Strengthening scientific integrity in a changing landscape
As Canada navigates an increasingly complex scientific and technological landscape, it is important to ensure that SIPs remain responsive to new challenges. One such challenge is research security. Open collaboration and knowledge sharing are crucial for scientific progress, but it is also necessary to safeguard sensitive information and infrastructure. These should be considered complementary priorities, that together enable Canada to remain a leader and trusted partner in global science. SIPs can articulate the importance of this balance by ensuring that the responsible conduct of research in government includes considerations for safeguarding data and for security-conscious research practices.
Another key consideration is the role of scientific integrity in the age of artificial intelligence (AI). The rapid adoption and advancement of generative AI tools present enormous potential for scientific progress, but simultaneously introduce new challenges around transparency, reproducibility and accountability. SIPs should evolve to address expectations for the responsible use of AI in science and research. Indeed, other jurisdictions such as the European Union have added language to their research codes of conduct to reflect these new realitiesFootnote 23.
Over the past two years, the OCSA has been conducting consultations and developing a revised version of the Model Policy on Scientific Integrity to ensure that the policy continues to be fit for purpose in the coming years. This updated policy will include provisions on the appropriate engagement of Indigenous partners and use of Indigenous data and information in research, reflect lessons learned from implementation, incorporate feedback from the consultation process and respond to emerging priorities such as open science, research security and AI. In a rapidly evolving landscape shaped by emerging technologies and shifting geopolitics, SIPs must remain nimble and forward-looking.
Canada’s ambition to become the “world’s leading hub for science and innovation”Footnote 24 needs to go hand-in-hand with a strong commitment to scientific integrity. Upholding the responsible conduct, communication and use of science is essential for maintaining and increasing public trust, enabling high-quality research and ensuring that science informs policy effectively. Beyond updating the SIP for federal organizations, there is an opportunity for Canada to expand its commitment to scientific integrity. By championing a whole-of-society culture of scientific integrity, Canada could establish itself as a global leader for excellence in science and evidence-informed decision-making across sectors.
Appendix A: List of departments and agencies that are required to implement a scientific integrity policy
- 1. Agriculture and Agri-Food Canada
- 2. Canada Border Services Agency
- 3. Canadian Food Inspection Agency
- 4. Canadian Grain Commission
- 5. Canadian Heritage
- 6. Canadian Space Agency
- 7. Correctional Service of Canada
- 8. Crown-Indigenous Relations Northern Affairs Canada
- 9. Department of National Defence
- 10. Environment and Climate Change Canada
- 11. Fisheries and Oceans Canada
- 12. Global Affairs Canada
- 13. Health Canada
- 14. Impact Assessment Agency of Canada
- 15. Indigenous Services Canada
- 16. Infrastructure Canada
- 17. Innovation, Science and Economic Development Canada
- 18. Library and Archives Canada
- 19. National Research Council
- 20. Natural Resources Canada
- 21. Public Health Agency of Canada
- 22. Public Services and Procurement Canada
- 23. Royal Canadian Mounted Police
- 24. Statistics Canada
- 25. Transport Canada
Appendix B: List of the fifteen scientific integrity policy compliance measures (CMs)
- CM1: In what year did your departmental SIP come into effect?
- CM2: Has the DDAA notified employees about the SIP? (mSIP s.7.1.1.)
- CM3: Has the DDAA implemented additional procedures, policies, guidelines, tools, training or professional development opportunities in support of the SIP? (mSIP s.7.1.2.)
- CM4: Does the DDAA have a process for reporting and recording instances of policy conflict or incompatibility with the SIP? (mSIP s. 3.5)
- CM5: Does the DDAA require that all technical communications undergo peer-review? (mSIP s.7.5.6. (v))
- CM6: Does the DDAA require that, where appropriate, research or scientific projects involving humans be reviewed and approved by a Research Ethics Board (REB)? See the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans – TCPS 2 (2018) (mSIP s.7.8.1 (x))
- CM7: Has the DDAA taken steps to notify/inform contractors or extramural collaborators of the DDAA SIP and encourage them to comply with its articles? (mSIP s.7.1.3.)
- CM8: Has the DDAA appointed a Science Integrity Lead (SIL)? (mSIP s.7.2.2.2.)
- CM9: Does the DDAA have a process in effect for bringing forward and investigating breaches of scientific integrity? (mSIP s.7.2.2.3.)
- CM10: Does the DDAA have a mechanism and/or procedure in effect for soliciting researcher/scientist advice on DDAA policy and/or regulations? (mSIP s.7.7.1.)
- CM11: Does the DDAA have a mechanism and/or procedure in effect for soliciting researcher/scientist advice on DDAA research programs? (mSIP s.7.7.2.)
- CM12: Does the DDAA have a mechanism and/or procedure in effect for systematically soliciting researcher/scientist assistance in identifying and prioritizing federal investment in research? (mSIP s.7.7.3.)
- CM13: Does the DDAA have measures in effect to support education, training and/or professional development in any of the following areas: responsible conduct in research; research ethics; and the annotation, management and archiving of research and scientific data? (mSIP s. 7.2.1.3.)
- CM14 (added in the 2022 survey and onward): Does your DDAA have a monitoring plan in effect for the DDAA SIP that will provide information on the extent to which the policy has achieved its objectives (policy performance)? (mSIP s.7.9)
- CM15 (added in the 2023 survey and onward): Does your DDAA have measures in place to support education, training and/or professional development devoted to the roles of science and research in developing evidence to support evidence-informed decision-making? (mSIP s.7.7.4)
Appendix C: Summary of initiatives advancing the objectivesFootnote 25 of the scientific integrity policy, as reported by departments and agencies
| OBJECTIVE 1: FOSTER A CULTURE OF SCIENTIFIC INTEGRITY |
OBJECTIVE 2: BUILD TRUST IN SCIENCE AND RESEARCH |
OBJECTIVE 3: CLARIFY EXPECTATIONS FOR SCIENCE CONDUCT |
OBJECTIVE 4: UNDERSTANDING ROLE OF SCIENCE IN DECISION-MAKING |
|
|---|---|---|---|---|
| EMPLOYEE SCIENTIFIC INTEGRITY AND ETHICS TRAINING | ✔ | ✔ | ✔ | ✔ |
| EMPLOYEE AWARENESS AND COMMUNICATIONS | ✔ | ✔ | ✔ | ✔ |
| EMPLOYEE WORKSHOPS AND DISCUSSION FORUMS | ✔ | ✔ | ✔ | ✔ |
| EXTERNAL/COLLABORATOR AWARENESS OF SIP AND RELATED GUIDELINES/DIRECTIVES | ✔ | ✔ | ||
| POLICY INSTRUMENTS AND GOVERNANCE FOR SCIENCE, SCIENCE ADVICE, AND DECISION-MAKING | ✔ | ✔ | ✔ | ✔ |
| POLICY REVISIONS AND ALIGNMENT, AND SIP IMPLEMENTATION GUIDELINES | ✔ | ✔ | ✔ | |
| SCIENTIFIC INTEGRITY IN THE CONTEXT OF PUBLIC SERVICE VALUES AND ETHICS, AND ITS INTEGRATION INTO DEPARTMENTAL CODES | ✔ | ✔ | ✔ | |
| INTERDEPARTMENTAL COLLABORATIONS TO ADVANCE SIP IMPLEMENTATION | ✔ | |||
| WORKING GROUPS/FOCUS GROUPS ON SCIENTIFIC INTEGRITY AND SIP IMPLEMENTATION | ✔ | ✔ | ✔ | ✔ |
| FAIR, TRANSPARENT AND TIMELY PROCEDURE FOR BRINGING FORWARD ALLEGATIONS OF BREACHES | ✔ | ✔ | ✔ | |
| APPOINTING SCIENTIFIC INTEGRITY CHAMPIONS | ✔ | |||
| PUBLIC ENGAGEMENT ON OR COMMUNICATION OF DEPARTMENTAL SIP OR SCIENCE/RESEARCH ACTIVITIES | ✔ | ✔ | ✔ | |
| ADMINISTRATIVE SUPPORT MECHANISMS TO PLAN, TRACK, AND OVERSEE DEPARTMENTAL SCIENCE AND RESEARCH (IN ALIGNMENT WITH SIP) | ✔ | ✔ | ||
| ETHICS REVIEW OF DEPARTMENTAL SCIENCE AND RESEARCH | ✔ | ✔ | ||
| APPLYING SCIENTIFIC INTEGRITY VALUES TO ALL AREAS OF KNOWLEDGE | ✔ | |||
| ENGAGEMENT WITH EXTERNAL EXPERTS AND THE SCIENCE/RESEARCH COMMUNITY | ✔ | ✔ |
